Prevention Strategies for Women with BRCA1 or BRCA2 Mutations
Given the uncertain utility of screening programs (secondary prevention) in this high-risk population, some women consider undergoing surgical procedures in an attempt to manage their risk. Bilateral risk-reducing mastectomy appears to be approximately 90% effective in reducing breast cancer incidence and mortality in women with a significant familial risk.
Unfortunately, failure can occur as the result of the development of breast cancer in residual breast tissue. This is a particular issue after subcutaneous mastectomy, but even women undergoing total (simple) mastectomy may develop cancer in ectopic rests of breast tissue (e.g., in the axilla). The effectiveness of mastectomy in women with germline BRCA mutations has not been defined. Despite its presumed effectiveness, the uptake of risk-reducing mastectomy by such women in the United States appears to be quite low.
Laparoscopic risk-reducing salpingo-oophorectomy is performed more commonly, particularly in women who have completed child-bearing and are nearing menopause. There are few data regarding the effectiveness of this procedure in reducing ovarian cancer risk. Oophorectomy cannot be considered completely protective, as cases of primary peritoneal carcinoma have been described after surgery.
Nevertheless, because of uncertainties regarding the effectiveness of ovarian cancer screening and the perceived high case-fatality rate of ovarian cancer, a significant fraction of women at defined hereditary risk choose to undergo the procedure. On a policy level, two separate decision analyses have demonstrated that risk-reducing surgery (mastectomy and/or oophorectomy) is a cost-effective way to manage cancer risk in this population. While these analyses are encouraging, it is difficult to translate these population benefits in a meaningful way to an individual patient.
There has been considerable interest in the use of chemoprevention as a means to reduce cancer risk. In particular, the encouraging results of the NSABP-P1 trial have provided a springboard for investigating the use of antiestrogens in BRCA families. At this time, it is unknown whether this strategy will be as effective as it is in the wider population.
Of particular concern is the fact that many BRCA-associated breast cancers are hormone-receptor negative, and neither tamoxifen nor raloxifene substantially affect the incidence of such tumors. Further analysis of the P1 study is ongoing to determine the impact of antiestrogen therapy on breast cancer risk in BRCA heterozygotes. Until these results are available, the use of tamoxifen and other antiestrogens should be considered investigational in this group.
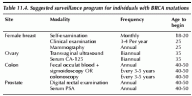
Table 11.4. Suggested surveillance program for individuals with BRCA mutations
Oral contraceptives have been known for some time to reduce ovarian cancer risk in the general population. A recent study has indicated a similar beneficial effect in BRCA heterozygotes. Specifically, heterozygotes who took oral contraceptives for any period of time had a relative risk for ovarian cancer of 0.5 (95% CI 0.3-0.8) when compared to controls. Benefit seemed to accrue particularly to women who took oral contraceptives for over 3 years.
Although the results of this study are encouraging, methodological limitations mandate confirmation of the results before these agents can be routinely recommended to women with BRCA mutations. This is particularly the case as the impact on breast cancer risk was not described in the aforementioned study, and at least one publication has suggested an increased risk of breast cancer among heterozygotes taking oral contraceptives when compared to controls.
Mark E. Robson
Breast conservation therapy for invasive breast cancer in Ashkenazi women with BRCA gene founder mutations. J Natl Cancer Inst 2003