Health Centers > Cancer Health Center > Overview of Cancer
Overview of Cancer - GENERAL
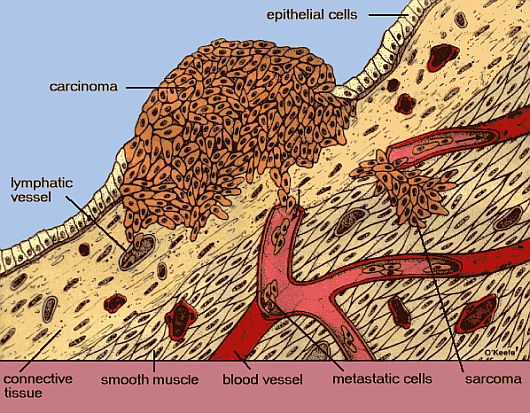
Cancer: A proliferation of cells whose unique trait - loss of normal controls - results in unregulated growth, lack of differentiation, local tissue invasion, and metastasis.
Cancer (malignancy) can develop in any tissue of any organ at any age. Most cancers are potentially curable if detected at an early stage. By performing self-examinations, patients can help recognize early signs of some possible malignancies. Diagnostic testing and therapy are essential for optimal results. When cure or reasonable palliation is likely, physicians must discuss all therapeutic options.
Patients with cancer that is unlikely to be cured need to be informed about what treatment is likely to accomplish and the side effects it may produce. Intensive care may be needed for treatment-related complications. Psychologic support from the physician and the health care team (which may include a psychiatrist and a social worker) helps patients through therapy (see Complications, below).
Physicians must be truthful, yet convey a sense of optimism. Some patients may need to be advised against health care practitioners who falsely promise cure. The patient should feel that members of the health care team are concerned and available to answer questions. The physician should initiate frank discussions about end-of-life care and advance medical directives at an appropriate time.
Cellular Kinetics
Generation time is the time it takes for cells to enter the cell cycle (see Fig. 142-1) and give rise to two daughter cells. Malignant cells usually have a shorter cycle than nonmalignant cells. Most nonmalignant cells have a larger percentage of cells in G0 (resting phase), so that there is a smaller proliferation fraction. Initial exponential tumor growth is followed by a plateau phase when cell death equals the rate of formation of daughter cells. Compared with large tumors, small tumors have a greater percentage of cells in cycle and thus greater proliferation.
Cellular kinetics are important in the design of antineoplastic drug regimens. Many antineoplastic drugs are effective only if cells are in cell cycle, and some drugs work only during a specific phase of the cycle. Cellular kinetics may influence the dosage schedules and timing of treatment.
Tumor Growth and Metastasis
As a tumor grows, nutrients are provided by direct diffusion from the circulation. Local tissue invasion can result in pressure on normal tissues, which can lead to inflammation, or the tumor may produce substances (eg, collagenase) that lead to enzymatic destruction of tissues. Subsequently, synthesis of tumor angiogenesis factor causes formation of an independent vascular supply to the tumor. Almost from inception, a tumor may shed cells into the circulation.
From animal models, it is estimated that a 1-cm tumor sheds > 1 million cells/24 h into the venous circulation. In animals, circulating tumor cells usually die as a result of intravascular trauma; the longer a tumor cell spends in the circulation, the greater the chance of its death. The probability that a circulating tumor cell will become a metastatic tumor is estimated at < 1:1 million.
Metastases develop when tumor cells adhere to vascular endothelium and penetrate into surrounding tissues, surviving and spawning independent tumors at distant sites. Thus, tumor growth resumes, disrupting normal tissue and organ function. Metastatic tumors can give rise to other metastases.
Experiments suggest that metastasis is not a random event and that the primary tumor may regulate the growth of metastatic tumors (eg, in renal cell carcinoma, the rate of growth is often similar in the primary and metastatic nodules). Theoretically, removal of the primary tumor can result in rapid growth of the metastases.
Molecular Abnormalities
Mutations in genes are partially responsible for the growth or reproduction of malignant cells. These mutations alter the quantity or behavior of the proteins encoded by growth-regulating genes and alter cell division. Two major categories of mutated genes are oncogenes and tumor suppressor genes.
Oncogenes are abnormal forms of the genes that normally regulate cell growth. For example, the ras gene is abnormal in about 25% of human cancers. The Ras protein (encoded by the ras gene) regulates or signals cell division. In most situations the gene is inactive, but in these malignant cells the Ras protein is active and signals cells to divide, even though they should not.
Another example of oncogene activity involves protein kinases, enzymes that help regulate many cellular activities, particularly signaling from the cell membrane to the nucleus, thus initiating the cell's entrance into the cell cycle and controlling several other functions. Several human cancers (eg, bladder cancer, breast cancer, chronic myelocytic leukemia [CML]) contain structurally altered protein kinase enzymes. When overproduced or altered, the kinase stimulates cell division continuously.
Cellular oncogenes are amplified in several human malignancies (eg, C-myc and N-myc in small cell lung cancer, N-myc in neuroblastoma, C-erb B-2 in breast cancer). The activation of oncogenes is not entirely understood, but many factors may contribute, including chemical carcinogens (eg, in tobacco smoke) or infectious agents (eg, viruses).
Activation may result from chromosomal (DNA) rearrangements. In Burkitt's lymphoma, t(8;14) moves the C-myc locus on chromosome 8 to a position distal to the immunoglobulin heavy chain locus on chromosome 14, increasing immunoglobulin synthesis. In CML, the t(9;22) results in a chimeric protein--a fusion of the N terminal portion of bcr and the C terminal portion of abl. These translocations of DNA are located near or on genes responsible for cell growth and proliferation.
There are several general categories of cancer, with carcinomas and, more specifically, adenocarcinomas being the most common:
- Carcinomas. Cancers that occur in epithelial surfaces; the cells that form the outer surface of the body to line or cover the body's cavities, tubes, and passageways.
- Adenocarcinomas. Cancers that form on a glandular surface, such as the lung, breast, prostate, ovary, or kidney. Adenocarcinomas are considered a subtype (specific type) of carcinomas.
- Sarcomas. Cancers that occur in supporting structures, such as bone, muscle, cartilage, fat, or fibrous tissue.
- Leukemias. Cancers that occur in blood cell elements, such as bone marrow.
- Lymphomas. Cancers that are found in cells of the immune system.
What are primary cancers?
Cancers begin in a single cell, and that cell is the site of the primary cancer. The cancer is named for the primary site of origin, such as skin, colon, or breast. For example:
- When cancer that started in the colon is found in the liver, it is called colon cancer that has metastasized to the liver, not liver cancer.
- Lung cancers are those that started from a lung cell. If it spreads to the brain, it is still lung cancer, not brain cancer.
- When cancer spreads to the regional lymph nodes, those nodes are said to contain metastatic cancer. (Cancers that originate in the lymph cells of a node are called lymphomas.)
What are metastatic cancers?
Cancer can spread from where it started to other parts of the body.
- Spreading may occur by direct extension or invasion into nearby tissues.
- Systemic spread throughout the body may occur by way of the cancer cell getting into and travelling through the:
- Blood system. Arteries and veins take blood to and from all areas of the body.
- Lymphatic system. A network of lymphatic vessels in all areas of the body that drain and filter infectious agents.
- Cerebrospinal fluid
When cancer spreads to another part of the body, it is called metastatic cancer.
Tumor suppressor genes normally suppress the development of malignancies by encoding for proteins that suppress tumor initiation and growth. For example, the retinoblastoma (RB) gene encodes for the protein pRB, which regulates the cell cycle by stopping DNA replication. Mutations in the RB gene occur in 30 to 40% of all human cancers, allowing affected cells to divide continuously.
Another important regulatory protein, p53, prevents replication of damaged DNA in normal cells and promotes cell death (apoptosis) in cells with abnormal DNA. Inactive or altered p53 allows cells with abnormal DNA to survive and divide. Mutations are passed to daughter cells, conferring a high probability of neoplasm. The p53 gene appears defective in most human cancers.
Chromosomal abnormalities:
Although phenotypic heterogeneity occurs with any malignancy, genotypically a given cancer is believed to arise from a clone of transformed cells. The factors that ultimately cause genic or chromosomal changes are unknown; however, the deletion, translocation, or duplication of important genes gives a cancer cell a proliferative advantage over normal cells, and a tumor may develop.
Chromosomal abnormalities are found in certain human cancers (see Table 142-1); eg, about 80% of patients with CML have the Philadelphia (Ph) t(9;22)chromosome. Using G6PD as a protein marker, only a single isoenzyme is present in RBCs and WBCs of patients with CML, whereas fibroblasts from these patients contain both isoenzymes. These findings indicate that a chromosomal change is present in the malignant cells. The loss of alleles located on chromosomes 17p and 18q appears important in the etiology of colorectal cancer. The loss of alleles on chromosome 17p has also been implicated in breast cancer, gliomas, lung carcinoma, and osteosarcoma. The sites 17p and 18q have been suggested as the location of tumor suppressor genes.
A chromosomal mechanism of the conversion of normal epithelium to cancer has been proposed for familial polyposis of the colon. First, loss of a suppressor gene on chromosome 5 makes the epithelium hyperproliferative. A DNA methylation change results in an early adenoma that ras oncogene converts to an intermediate adenoma; loss of a suppressor gene on chromosome 18 converts it to a late adenoma, and loss of a gene on chromosome 17 converts it to cancer. Other genetic changes may be required for the cancer to metastasize.
Chromosomal analysis of cancer cells may provide prognostic or therapeutic information; eg, patients with acute myelogenous leukemia and normal chromosomes have a better prognosis than those with abnormal chromosomes. Similarly, patients with a translocation of chromosomes t(15;17) always develop acute promyelocytic leukemia.
In some congenital diseases, chromosomes break easily, putting children at high risk of developing acute leukemia and other cancers. These include Bloom's syndrome (a rare autosomal recessive disorder with dwarfism, a photosensitive telangiectatic facial erythema, and characteristic facies), Fanconi's syndrome, and Down syndrome (trisomy 21).
Environmental Factors
Viruses linked with human malignancies include human papillomaviruses (cervical carcinoma), cytomegalovirus (Kaposi's sarcoma), Epstein-Barr virus (Burkitt's lymphoma, immunoblastic lymphoma, and nasopharyngeal carcinoma), and hepatitis B virus (hepatocellular carcinoma). Human retroviruses have been linked to T-cell lymphomas (human T-cell lymphotrophic virus [HTLV-1]), which have a predilection for skin and bone involvement, hypercalcemia, and a leukemic phase. The mechanism of HTLV-1 neoplastic transformation is integration of the provirus (double-stranded DNA copy of the viral RNA genome) into the cellular genome. HTLV-2 has been tenuously associated with an unusual form of hairy cell leukemia of a T-cell linkage. HIV types I and II are pathogenically linked to AIDS. Patients with AIDS are predisposed to Kaposi's sarcoma and lymphoma, which may be viral in origin.
Of parasites, Schistosoma haematobium has been linked to bladder cancer, which usually develops after chronic inflammation and fibrosis. Opisthorchis sinensis has been linked to carcinoma of the pancreas and bile ducts.
Chemical carcinogenesis is a multistep process: (1) In initiation, a cell that experiences a carcinogenic event can potentially develop into a neoplastic clone. (2) In promotion, which is reversible, continued existence of the neoplastic clonal proliferation depends on a chemical or agent with little carcinogenic activity. (3) In progression, irreversible growth of altered (neoplastic) cells occurs. An agent that possesses little or no carcinogenic potency (a cocarcinogen) enhances the carcinogenic effect of another agent when exposure is simultaneous. Chemical carcinogenesis is influenced by age, endocrine status, diet, other exogenous agents (cocarcinogens or promoters), and immunologic status. Common chemical carcinogens are listed in Table 142-2.
Ultraviolet radiation is a definite cause of skin cancer (ie, basal and squamous cell carcinoma, melanoma, and especially in xeroderma pigmentosum).
Ionizing radiation is carcinogenic; eg, survivors of the atomic bomb drop in Hiroshima and Nagasaki have a higher-than-expected incidence of leukemia and several other cancers. Similarly, when ionizing radiation in the form of x-rays is used to treat nonmalignant disease (acne, thymic or adenoid enlargement, and ankylosing spondylitis), the incidence of cancer increases, including acute and chronic leukemias; Hodgkin's and non-Hodgkin's lymphomas; multiple myeloma; aplastic anemia terminating in acute nonlymphocytic leukemia (ANLL); myelofibrosis; melanoma; and thyroid cancer. Industrial exposure (eg, to uranium by mine workers) is linked to development of lung cancer after a 15- to 20-yr latency. Long-term exposure to occupational irradiation or to internally deposited thorium dioxide predisposes persons to angiosarcomas and ANLL.
Chronic irritation to skin leads to chronic dermatitis and on rare occasions ultimately to squamous cell carcinoma.
Immunologic Disorders
Patients with immunologic disorders are predisposed to lymphoreticular neoplasia and should be screened periodically; development of new or suspicious lymphadenopathy should be evaluated by biopsy. In patients with ataxia-telangiectasia, the incidence of acute lymphoblastic leukemia (ALL), brain tumors, and gastric cancer exceeds that of the normal population. Patients with Wiskott-Aldrich syndrome and X-linked agammaglobulinemia are also at high risk for lymphoma and ALL.
Patients with immune deficiency, either as a result of immunosuppressive drugs or HIV infection, are at risk for various neoplasms, especially large cell lymphoma and Kaposi's sarcoma. Similarly, patients with SLE, RA, or Sjogren's syndrome are at risk for lymphoma, usually the B-cell type, presumably related to the altered immunologic status.
Diagnosis and Screening
A complete history and physical examination are prerequisites to early diagnosis. Physicians must be aware of predisposing factors and must specifically ask about familial cancer, environmental exposure, and prior illness (eg, autoimmune diseases, previous immunosuppressive therapy, AIDS). The review of systems is important and should address symptoms of fatigue, weight loss, fevers or night sweats, cough, hemoptysis, hematemesis or hematochezia, change in bowel habits, and persistent pain. The physical examination should direct particular attention to skin, lymph nodes, lungs, breasts, abdomen, and testes and to the prostate, rectal, and vaginal examinations.
The principal objectives of cancer screening and early diagnosis are to decrease cancer mortality, allow use of less radical therapy, and reduce financial costs. Screening procedures that have decreased cancer mortality are the Papanicolaou (Pap) smear (cervical cancer) and breast self-examination and mammography (breast cancer). For screening procedures recommended by the American Cancer Society, see Table 142-3. However, screening has its own risks: physical and psychologic morbidity from false-positive results, false reassurance for patients with false-negative findings, and morbidity from the screening procedure.
What are diagnostic procedures for cancer?
When symptoms suggest cancer, your doctor may request or perform any of the following procedures to help diagnose it:
- A detailed medical history - family and personal
- Thorough physical exam
- Pelvic exam of the uterus, vagina, ovaries, bladder, and rectum (women only)
- Pap test at the time of pelvic exam (women only)
- Rectal exam of the prostate and rectum (men only)
Other diagnostic procedures that may be requested include:
Imaging tests, such as:
- X-ray
- Computed tomography (CT or CAT scan). A noninvasive diagnostic imaging procedure that uses a combination of X-ray and computer technology to produce detailed images (often called slices) of the body. The CT scan may show tumors or indicate enlarged lymph nodes--a possible sign of a spreading cancer or of an infection.
- Radionuclide or nuclear medicine scan. An imaging scan in which a small amount of radioactive substance is injected into the vein. A machine measures levels of radioactivity in certain tissues or organs, thereby detecting any abnormal areas or tumors. Some examples are bone scans, PET scans, thyroid scans, and gallium scans.
- Ultrasound. An imaging technique that uses high-frequency sound waves to produce an image on a monitor of the abdominal organs, such as the uterus, liver, and kidneys.
- Magnetic resonance imaging (MRI). A noninvasive procedure that produces detailed views of an internal organ or structure, especially the brain and spinal cord, without the use of X-rays. The MRI may show abnormal nodules in bones or lymph nodes - a sign that cancer may be spreading.
- Endoscopy. Use of a flexible tube with a lens or tiny camera (and a light on the end), which is connected to a computer screen, allowing the doctor to see inside the hollow organs, such as the esophagus, stomach, intestines, bladder, or uterus. Biopsy samples (tiny pieces of tissue) can be taken through the tube for further evaluation.
- Lab tests. These are done to examine blood, urine, other fluids, or tumor tissue from a biopsy.
- Biopsy. This is done to remove a sample of the suspicious tissue for examination in a lab by a pathologist. Biopsy can be done with several different procedures depending on the location of the tumor and size. Endoscopy, needle biopsy with image guidance, and surgical biopsy are a few examples.
For most types of cancer, exams and imaging tests might suggest a person has cancer, but a biopsy is usually needed to be sure of the diagnosis.
Once the cancer is diagnosed, an evaluation will be made to determine the extent (stage) of the cancer. This is often done with some of the imaging tests described above.
Tumors may synthesize proteins that produce no clinical symptoms, eg, β-human chorionic gonadotropin, α-fetoprotein, carcinoembryonic antigen, CA 125, and CA 153. These protein products may be used as tumor markers in the serial evaluation of patients for determining disease recurrence or response to therapy.
Staging
Once a histologic diagnosis is made, staging (ie, determination of the extent of disease) helps determine treatment decisions and prognosis. Clinical staging uses data from the patient's history, physical examination, and noninvasive studies. Pathologic staging requires tissue specimens. For staging of specific neoplasms, see details elsewhere in The Manual.
Mediastinoscopy is especially valuable in the staging of non-small cell lung cancer; if it shows mediastinal lymph node involvement, the patient would not usually benefit from a thoracotomy and lung resection.
Bone marrow biopsy is especially useful in determining metastases from malignant lymphoma and small cell lung cancer. Marrow biopsy will be positive in 50 to 70% of patients with malignant lymphoma (low and intermediate grade) and in 15 to 18% of patients with small cell lung cancer at diagnosis.
Axillary lymph node removal with histopathologic examination for metastases (part of surgical therapy) helps in staging breast cancer.
Laparotomy in colon carcinoma allows for therapeutic intervention and operative staging. Laparotomy with splenectomy, lymph node removal, and liver biopsy is an integral part of the staging of Hodgkin's disease in certain patients.
Serum chemistries and enzymes may help staging. Elevation of liver enzymes (alkaline phosphatase, LDH, and ALT) suggests the presence of liver metastases. Elevated alkaline phosphatase and serum Ca may be the first evidence of bone metastases. Elevated acid phosphatase (tartrate inhibited) suggests extracapsular extension of prostate cancer. Fasting hypoglycemia may indicate an insulinoma, hepatocellular carcinoma, or retroperitoneal sarcoma. Elevated BUN or creatinine levels may indicate an obstructive uropathy secondary to a pelvic mass, intrarenal obstruction from tubular precipitation of myeloma protein, or uric acid nephropathy from lymphoma or other cancers. Elevated uric acid levels often occur in myeloproliferative and lymphoproliferative disorders. α-Fetoprotein may be elevated in hepatocellular carcinoma and testicular carcinomas, carcinoembryonic antigen-S in colon cancer, β-human chorionic gonadotropin in choriocarcinoma and testicular carcinoma, serum immunoglobulins in multiple myeloma, and DNA probes (bcr probe to identify the chromosome 22 change) in CML.
Imaging studies, especially CT and MRI, can detect metastases to brain, lung, spinal cord, or abdominal viscera, including the adrenal glands, retroperitoneal lymph nodes, liver, and spleen. MRI (with gadolinium) is the procedure of choice for recognition and evaluation of brain tumors.
Ultrasonography can be used to study orbital, thyroid, cardiac, pericardial, hepatic, pancreatic, renal, and retroperitoneal areas. It may guide percutaneous biopsies and differentiate renal cell carcinoma from a benign renal cyst. Lymphangiography reveals enlarged pelvic and low lumbar lymph nodes and is useful in the clinical staging of patients with Hodgkin's disease, but it has generally been replaced by CT.
Liver-spleen scans can identify liver metastases and splenomegaly. Bone scans are sensitive in identifying metastases before they are evident on x-ray. Because a positive scan requires new bony formation (ie, osteoblastic activity), this technique is useless in neoplasms that are purely lytic (eg, multiple myeloma); routine bone x-rays are the study of choice in such diseases. Gallium scans can help in staging lymphoid neoplasms. Radiolabeled monoclonal antibodies (eg, to carcinoembryonic antigen, small cell lung cancer cells) provide important staging data in various neoplasms (eg, colon cancer, small cell lung cancer).
Complications
Malignancy may lead to pain, wasting, neuropathy, nausea, anorexia, seizures, hypercalcemia, hyperuricemia, obstruction, and organ failure.
Cardiac tamponade often occurs precipitously. The most common causes are breast and lung cancer and lymphoma. Because a malignant pericardial effusion precedes cardiac tamponade, patient history usually reveals ill-defined chest pain or pressure that is worse when supine and better when sitting up. For diagnostic and therapeutic purposes, a pericardiocentesis must be done and a pleuropericardial window or pericardiectomy considered. Pleural effusions, if present, should be drained if symptomatic and followed for reaccumulation. If the effusion reaccumulates rapidly, thoracostomy tube drainage and sclerosing agents should be used. Spinal cord compression requires immediate attention to avoid morbidity. Hypercalcemia can be caused by malignancy. Superior vena cava syndrome, a dramatic clinical situation, requires urgent but not emergency care.
Pain in patients with metastatic cancer frequently results from bone metastases, nerve or plexus involvement, or pressure exerted by a tumor mass or effusion.
Paraneoplastic syndromes:
The paraneoplastic syndromes may be a result of excessive or ectopic hormones synthesized by a tumor, immune complexes, ectopic receptor production, release of physiologically active compounds, or unknown causes. Hormone production by tumor cells includes fasting hypoglycemia (insulin from insulinoma), diarrhea (vasoactive intestinal polypeptide from a neuroendocrine tumor, ie, islet cell tumor), and hypertension (epinephrine and norepinephrine from pheochromocytoma). Ectopic hormone synthesis includes ectopic ACTH and ADH (from small cell and non-small cell lung cancer), parathyroid hormone (from squamous cell lung cancer, head and neck cancer, bladder cancer), calcitonin (from breast cancer, small cell lung cancer, and medullary thyroid carcinoma), and thyroid-stimulating hormone (from gestational choriocarcinoma). Manifestations vary with the type of hormone produced. Successful treatment is best obtained by controlling the underlying malignancy, but symptoms can be palliated with drugs, eg, minocycline for ectopic ADH, cyproheptadine for carcinoid syndrome, or pamidronate and corticosteroids for hypercalcemia.
Clinical syndromes associated with malignancies with no identifiable products are truly paraneoplastic and may involve various organ systems. Neurologic paraneoplastic syndromes include subacute cerebellar degeneration, amyotrophic lateral sclerosis, sensory or sensory-motor peripheral neuropathy, Guillain-Barre syndrome, dermatomyositis, polymyositis, myasthenia gravis, and Eaton-Lambert syndrome. Most carcinomatous polyneuropathies are of unknown cause and have no specific therapy, although myasthenia gravis can be treated with neostigmine or prednisone.
Hematologic paraneoplastic syndromes are pure RBC aplasia, anemia of chronic disease, leukocytosis (leukemoid reaction), thrombocytosis, eosinophilia, basophilia, and disseminated intravascular coagulation. In addition, idiopathic thrombocytopenic purpura and a Coombs'-positive hemolytic anemia can complicate the course of patients with lymphoid malignancies and Hodgkin's disease.
Renal paraneoplastic syndrome (membranous glomerulitis) may occur in patients with colon cancer, ovarian cancer, and lymphoma as a result of circulating immune complexes.
Pigmented skin lesions or keratoses associated with malignancy include acanthosis nigricans (GI malignancy), generalized melanosis (lymphoma, melanoma, hepatocellular carcinoma), Bowen's disease (lung, GI, and GU malignancy), and large multiple seborrheic keratoses, ie, the sign of Leser-Trelat (lymphoma, GI malignancy).
Other miscellaneous paraneoplastic complications include fever, lactic acidosis (leukemia, lymphoma), hyperlipidemia (myeloma), and hypertrophic pulmonary osteoarthropathy (lung cancer or lung metastases from renal cancer, thymoma, sarcoma, and Hodgkin's disease).
METASTATIC CARCINOMA OF UNKNOWN PRIMARY ORIGIN
A biopsy-proven metastatic malignancy for which a primary site cannot be found.
Metastatic carcinomas of unknown primary origin constitute 0.5 to 7% of all cancers. Because cancer treatment often varies by tissue type, thorough evaluation is necessary. A detailed history and physical examination should be performed, with particular attention to signs and symptoms of breast, pelvic, prostate, rectal, and GI diseases. Laboratory tests should include a CBC, urinalysis, stool examination for occult blood, and serum chemistries (including prostate-specific antigen assays in males). X-rays should be limited to a chest x-ray, abdominal CT, and mammography. An upper GI series and barium enema should not be done routinely. On available cancerous tissue, immunoperoxidase staining for immunoglobulin, gene rearrangement studies, and electron microscopy help diagnose large cell lymphoma, whereas immunoperoxidase staining for α-fetoprotein or β-HCG may suggest germ cell tumors. Tissue analysis for estrogen and progesterone receptors helps identify breast cancer, and immunoperoxidase staining for prostate-specific antigen helps identify prostate cancer.
Even if a precise histologic diagnosis cannot be made, patients with poorly differentiated carcinomas near or at midline regions (eg, mediastinum, retroperitoneum) should be offered two cycles of a cisplatin-based regimen. If a response occurs, three to four cycles should be given, because nearly 50% of patients experience long disease-free intervals. For other patients, some physicians consider a trial with doxorubicin or paclitaxel-based regimens. In general, the responses are modest and of brief duration. However, because many patients harbor occult primary cancers, future management is likely to improve as more effective chemotherapy is developed for many tumor types.