NCCS pioneers new drug regimen which reduces toxicities for renal cancer patients
A study led by the Genitourinary (GU) oncology team at National Cancer Centre Singapore (NCCS) has revealed conclusive results in reducing toxicities for Asian patients with metastatic renal cell carcinoma (mRCC) or cancer that has spread beyond the kidney.
The seven-year study began in 2007 and the findings revolutionised the standard protocol for patient management in NCCS with an attenuated-dose regimen of sunitinib for patients with mRCC.
The new treatment regimen for sunitinib has been accepted by oncologists in Singapore. For the patients, this would mean an estimated 30 per cent reduction in fees because of the lower dosage. The median overall survival rate (OStotal) was 27.4 as compared to 21.8 months among patients receiving the attenuated dosage.
Sunitinib was introduced as a treatment for mRCC in Singapore since early 2005. The U.S. Food and Drug Administration (FDA) approved dosing of sunitinib is 50mg once daily for four weeks, followed by a two-week break in a six-week treatment cycle (conventional-doseregimen). Subsequent findings from 2005 to 2006 show that high toxicities were observed with the conventional dosing, especially in Asians.
“Many of the patients were experiencing severe side effects of grade 3 or higher with the conventional dosing. Our immediate response was to refine the treatment protocol to improve patients’ quality of life”, explained Dr Tan Min Han, Visiting Consultant, Division of Medical Oncology and member of the GU team, NCCS.
NCCS initiated a prospective clinical registry with 127 mRCC patients receiving attenuated sunitinib dosing of 37.5mg/d/4/2 (37.5mg of sunitinib once daily for four weeks, followed by a two-2 week break) as treatment protocol in 2007. Clinical data of patients receiving sunitinib at NCCS from 2005 to 2012 and three other tertiary centres in Singapore (Johns Hopkins-International Medical Centre, National University Hospital Singapore, and Onco-Care of Gleneagles Medical Centre) from 2005 to 2009 were used for comparison, representing at least 90 percent of all patients with mRCC treated over the period.
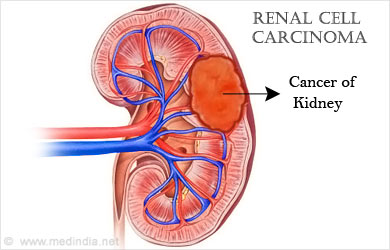
Kidney Cancer Facts
The American Cancer Society estimates that more than 58,240 people were diagnosed with kidney cancer in 2010. In the 1980s, up to 80% of kidney cancers were in advanced stages when they were diagnosed. Today, thanks to more advanced detection methods, only about 40% of cases are advanced at diagnosis.
You have two kidneys, one on each side of the back above the waist. Kidneys filter blood, and the waste is carried in the urine, which is produced by microscopic tubules. Urine flows into the ureter tubes and down into the bladder. Cancer arises from the microscopic tubules inside the kidney. Although kidney cancer usually grows as a single mass within the kidney, a kidney may contain more than one tumor, or tumors may be found in both kidneys.
Surgery offers the highest chance for successful treatment when kidney cancer has not spread. Once the cancer has spread beyond the kidney, the chance for successful treatment becomes much lower. Since 2006, new-generation drugs called targeted therapies have become available to control the cancer more successfully than prior medications.
Kidney Cancer Types
Renal cell carcinoma (cancer) (RCC) is the most prevalent form of kidney cancer. Types of RCC include clear cell, papillary, chromophobe and collecting duct carcinomas. Clear cell carcinoma accounts for 80% of all RCC cases, and most treatments are focused on this type.
Wilms’ tumor is a childhood cancer, responsible for 95% of pediatric kidney cancer cases. Learn more about Wilms’ tumor.
Urothelial cancer of the kidney pelvis and ureter: Cancer of the urinary tract that occurs in the kidney or ureter is called urothelial carcinoma. It is not considered kidney cancer, although it is frequently called that in error.
Kidney cancer is cancer that originates in the kidneys. Your kidneys are two bean-shaped organs, each about the size of your fist. They’re located behind your abdominal organs, with one kidney on each side of your spine.
In adults, the most common type of kidney cancer is renal cell carcinoma. Other less common types of kidney cancer can occur. Young children are more likely to develop a kind of kidney cancer called Wilms’ tumor.
The incidence of kidney cancer seems to be increasing. One reason for this may be the fact that imaging techniques such as computerized tomography (CT) scan are being used more often. These tests may lead to the accidental discovery of more kidney cancers.
Renal cell carcinoma (RCC) is the most common renal cancer. Symptoms can include hematuria, flank pain, a palpable mass, and FUO. However, symptoms are often absent, so the diagnosis is usually suspected based on incidental findings. Diagnosis is confirmed by CT or MRI and occasionally by biopsy. Treatment is with surgery for early disease and targeted therapy, an experimental protocol, or palliative therapy for advanced disease.
RCC, an adenocarcinoma, accounts for 90 to 95% of primary malignant renal tumors. Less common primary renal tumors include transitional cell carcinoma, Wilms tumor (most often in children - Wilms’ Tumor), and sarcoma.
In the US, about 65,000 cases of RCC and pelvic tumors and 13,000 deaths occur each year. RCC occurs slightly more often in men (male:female incidence is about 3:2). People affected are usually between 50 and 70 yr. Risk factors include the following:
Smoking, which doubles the risk (in 20 to 30% of patients)
Obesity
Excess use of phenacetin
Acquired cystic kidney disease in dialysis patients
Exposure to certain radiopaque dyes, asbestos, cadmium, and leather tanning and petroleum products
Some familial syndromes, particularly von Hippel - Lindau disease
RCC can trigger thrombus formation in the renal vein, which occasionally propagates into the vena cava. Tumor invasion of the vein wall is uncommon. RCC metastasizes most often to the lymph nodes, lungs, adrenal glands, liver, brain, and bone.
 The data revealed favourable results between the attenuated dosing regimen compared to the conventional dosing. 59 percent of the participants experienced severe side effects as compared to the previous 85 percent; 24 percent than 58 percent required reduction in dose delays; and 35 percent rather than 70 percent of patients requiring dose reduction during their course of treatment. Both dose delays and reduction are only required when high level of toxicities are observed.
The data revealed favourable results between the attenuated dosing regimen compared to the conventional dosing. 59 percent of the participants experienced severe side effects as compared to the previous 85 percent; 24 percent than 58 percent required reduction in dose delays; and 35 percent rather than 70 percent of patients requiring dose reduction during their course of treatment. Both dose delays and reduction are only required when high level of toxicities are observed.
Dr Tan reiterated the importance of the findings, “This is an affirmation to our efforts and we believed that the continuous understanding of real world outcomes will reap greater benefits for our patients. The findings would not be possible without the collaborative nature of our tertiary healthcare counterparts.”
###
 This research was published in Clinical Genitourinary Cancer journal in November 2014 and supported by a grant of $50,000 from the NCC Research Fund.
This research was published in Clinical Genitourinary Cancer journal in November 2014 and supported by a grant of $50,000 from the NCC Research Fund.
For more information, please contact:
National Cancer Centre Singapore
Corporate Communications Department
Rachel Tan
Tel: +65 6236-9535
Hp: +65 9754-0842
Email: .(JavaScript must be enabled to view this email address)
Edwin Yong
Tel: +65 6236-9465
Hp: +65 9116-6850
Email: .(JavaScript must be enabled to view this email address)
###
Ms. Rachel Tan
.(JavaScript must be enabled to view this email address)
65-623-69535
SingHealth
Journal
Clinical Genitourinary Cancer